A finished medicine pack can look correct from the outside and still fail a basic quality check. A tablet bottle may contain one less tablet than required. A carton may leave the cartoner without a leaflet. A saszetka may be slightly underfilled. A blister carton may miss one card while the outer box looks sealed and acceptable.
These errors are small in weight but large in consequence. They can create customer complaints, rework, batch investigation, and product loss. On fast packaging lines, visual checking alone is not enough. A checkweigher adds a weight-based inspection step that verifies each pack while it moves through production and removes units outside the accepted range.
For pharma packaging, the value is not limited to weighing. The same station can help confirm pack completeness across bottling lines, blister cartoning packing lines, sachet lines, and final cartons. The best result comes from choosing the right location, weight tolerance, reject method, and data setup for the product format.

What Is a Checkweigher?
A checkweigher is an automatic weighing system that checks products as they pass along a conveyor. In pharma packaging, it is commonly used to inspect filled bottles, saszetki, pouches, blister cartons, and retail cartons.
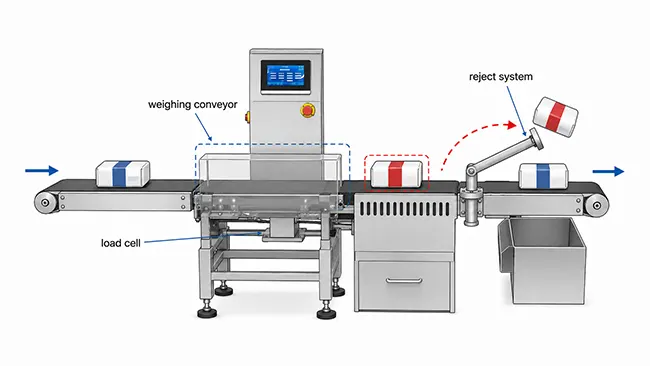
Most production lines use an inline checkweigher, also called a dynamic checkweigher. The pack does not stop during inspection. It enters the weighing section, the load cell captures the weight, and the control system compares that value with the approved upper and lower limits.
If the pack is within range, it continues downstream. If the pack is too light or too heavy, the reject system removes it from the line. The rejected unit may go into a locked bin, a reject chute, or a controlled collection area, depending on the product and quality procedure.
A static balance can still be useful for sampling, setup, and laboratory checks. A moving production line needs dynamic weighing because every pack must be inspected without stopping output.
Why Weight Errors Are a Packaging Quality Signal
Weight variation often points to a specific packaging problem.
A light tablet bottle may indicate a short count. A heavy bottle may contain extra tablets or an incorrect component. A carton with a small weight drop may be missing a leaflet, blister card, spoon, insert, or bottle. A sachet outside the target range may point to filling drift.
The checkweigher does not “see” the product like a vision system. It detects whether the pack matches the expected weight profile. That makes it useful when the missing item is hidden inside a closed bottle or carton.
In regulated production, automatic and electronic equipment used for drug manufacturing, processing, packing, or holding must be checked, inspected, or calibrated under written programs, with records kept. That makes routine verification and calibration part of proper checkweigher use, not only a maintenance task.
How an Inline Checkweigher Works
A stable weighing result starts before the pack reaches the load cell. Product spacing, conveyor transfer, vibration control, and pack orientation all affect accuracy.
The usual process is simple:
| Krok | What happens | Why it affects accuracy |
| Product infeed | Packs enter with controlled spacing | Prevents two packs from entering the weighing section too closely |
| Weighing conveyor | The pack crosses the load cell area | Captures the moving weight value |
| Weight comparison | Software checks the result against limits | Decides pass or fail |
| Odrzucenie | Failed packs are removed | Keeps defective units out of downstream flow |
| Data recording | Results are stored or reported | Supports batch review and troubleshooting |
A high speed checkweigher needs enough weighing time to produce a reliable result. If the conveyor is too short, the pack is unstable, or the line speed is pushed too high, the system may produce false rejects or miss borderline defects.
Where a Checkweigher Fits in Pharma Packaging Lines
The right position depends on the error you want to catch. One line may need to verify filled bottles before capping. Another may need to confirm finished cartons after leaflet insertion. A sachet line may need weight control right after sealing.
| Packaging format | Common risk | Useful checkweigher position |
| Tablet bottle | Missing tablets, extra tablets, abnormal filled bottle weight | After counting |
| Capsule bottle | Short count, empty bottle, abnormal component weight | After capsule counting |
| Sachet or pouch | Underweight or overweight fill | Po zapieczętowaniu |
| Blister carton | Missing blister card, missing leaflet | After cartoning |
| Final retail carton | Missing bottle, leaflet, spoon, insert, or leaflet pack | Before final collection or case packing |
For bottle packaging, checkweighing is often placed after counting. If the main risk is short-count bottles, an earlier position may allow faster rejection before more packaging value is added. If the goal is final pack verification, a later position may be better.
For blister carton lines, the unit is usually placed after cartoning. At that point, it can verify the combined weight of carton, blister cards, leaflet, and other inserted items.
Checkweigher vs Tablet Counter
A tablet counter controls how many tablets or capsules enter each bottle. A checkweigher verifies whether the filled pack matches the expected weight after filling or after another packaging step.
They are not substitutes. They protect different points in the line.
A tablet counter handles the counting action. The weighing station checks the outcome. If a bottle is short-counted, overfilled, empty, or outside the normal range, the checkweigher can reject it before the issue moves into sealing, labeling, or cartoning.
This second inspection layer is useful because pack weight can change for reasons beyond tablet count. Bottle weight variation, cap weight, desiccant presence, label material, leaflet weight, or carton board variation may affect the final result. The buyer should decide whether the inspection target is product count, finished bottle completeness, or final carton completeness before choosing the location.

Common Problems a Checkweigher Can Catch
A pharma packaging line may use weight inspection to control several common risks.
Short-count bottles
A bottle filled with fewer tablets or capsules than required may still look normal. A weight check can flag the bottle if the missing quantity creates a detectable difference.
Overfilled bottles
Extra tablets or capsules can create label claim and quality problems. The same system can remove packs above the accepted limit.
Underweight sachets
Powder and granule sachet lines need fill control after sealing. A checkweigher can reject packs that fall below the fill range.
Incomplete cartons
Closed cartons can hide missing leaflets, blister cards, spoons, or bottles. Weight checking gives a non-opening method to screen finished units.
Filling drift
A steady movement toward the low or high side of the range may point to upstream filling instability. Early detection reduces scrap and rework.
Component mix issues
If different cartons, butelki, caps, or leaflets are used by mistake, the weight profile may move outside the expected range.
What Causes False Rejects?
False rejects waste good product and slow the line. They often come from line conditions rather than the weighing cell itself.
Common causes include unstable bottle spacing, packs touching each other, vibration from nearby machines, poor transfer between conveyors, luźne czapki, tilted cartons, product shaking inside containers, and overly tight weight limits.
Reject timing is another frequent source of trouble. If the system detects a failed pack but the reject device fires too early or too late, the wrong unit may be removed. This becomes a serious control issue in pharma production because rejected products must be traceable and properly handled.
Good setup reduces these problems. The checkweigher conveyor should match the pack size and line speed. The infeed should separate products consistently. Weight limits should reflect actual material variation, not only theoretical pack weight. The reject station should include confirmation sensors when required by the quality plan.
How to Choose a Checkweigher for Pharma Packaging
The best machine is not always the one with the highest claimed accuracy. The better question is whether it can hold stable accuracy at your normal production speed, with your pack shape, material variation, and line layout.
Buyers should check these points before selection:
| Buying point | What to check |
| Product format | Butelka, sachet, carton, blister carton, kieszonka, lub produkty mieszane |
| Weight range | Minimum and maximum pack weight |
| Accuracy at speed | Dynamic accuracy under production conditions |
| Conveyor design | Length, belt width, transfer stability, and spacing control |
| Reject method | Air jet, pusher, diverter, flap, or custom rejection |
| Reject confirmation | Sensor confirmation and controlled reject collection |
| Data needs | Batch reports, reject logs, user access, and audit trail |
| Cleaning access | Open design, material choice, and GMP-style surfaces |
| Integracja | Fit with counting, capping, opieczętowanie, labeling, and cartoning machines |
For electronic records used in regulated environments, the FDA’s Part 11 guidance is often reviewed when systems manage electronic records or signatures required by predicate rules. Buyers should confirm record handling, user authority, audit trails, and data export requirements during the equipment selection stage.
Checkweigher, Metal Detector, and Vision Inspection
Weight inspection does not replace every inspection device.
A checkweigher detects weight-based problems. It is suitable for missing components, wrong counts, underweight packs, overweight packs, and incomplete cartons.
A metal detector looks for metallic contamination. It cannot confirm that a carton contains the right leaflet or that a bottle has the correct number of capsules.
A vision inspection system checks visible features, such as label position, printed code, barcode quality, cap presence, carton flap condition, or product appearance before closing.
Some lines combine these technologies. A metal detector checkweigher can save floor space and reduce transfer points. A vision system after labeling can check print and label quality. The right combination depends on the defect types, available space, validation needs, and budget.
How Weight Inspection Fits the Full Line
For tablet and capsule products, the checkweigher is usually part of a wider packaging route rather than a standalone decision. It may need to connect with a tablet counting and bottling line, Maszyna zliczania kapsułek, bottle capping machine, Maszyna uszczelniająca indukcyjna, maszyna do etykietowania, or cartoning machine.
Ruidapacking’s equipment range covers solid dosage production and back-end packaging, so line planning can consider both upstream tablet or capsule handling and downstream counting, blistering, labeling, and cartoning. This is useful when weight inspection needs to fit an existing line or when a new line must be planned around bottle, pęcherz, sachet, or carton formats.

Wniosek
A checkweigher helps pharma packaging lines find problems that are easy to miss at normal production speed. A small weight change can point to a short-count bottle, underweight sachet, missing blister card, absent leaflet, or incomplete carton.
Good results depend on more than the weighing specification. The line needs stable product spacing, suitable conveyor design, realistic weight limits, reliable rejection, and useful records. Buyers should choose the inspection position according to the defect they need to catch, not only the available floor space.
For pharma packaging, weight inspection works best when it is planned as part of the full line. Rachunkowość, pożywny, capping, opieczętowanie, labeling, cartoning, and final inspection all affect whether the finished pack is complete, controlled, and ready to ship.
Często zadawane pytania
What is a checkweigher used for in pharma packaging?
It weighs each pack on the line and removes products outside the approved weight range. It can help detect short-count bottles, underweight sachets, missing leaflets, missing blister cards, and incomplete cartons.
Is a checkweigher the same as a tablet counter?
NIE. A tablet counter controls how many tablets or capsules enter each bottle. A checkweigher verifies the weight of the filled or finished pack after the counting or packaging step.
Can a checkweigher detect one missing tablet?
Tak, if the missing tablet creates a measurable weight difference and the normal variation of bottles, caps, labels, and product is controlled. Very light tablets or wide material variation may require tighter setup and testing.
Why does a checkweigher reject good products?
False rejects can come from vibration, unstable spacing, tilted packs, luźne czapki, product shaking, narrow weight limits, or incorrect reject timing.
What is an inline checkweigher?
An inline checkweigher weighs products while they move through the production line. It supports continuous inspection without stopping each pack.
Do all pharma lines need a metal detector checkweigher?
NIE. A metal detector checkweigher is useful when the line needs both weight inspection and metal contamination detection in one unit. Some lines use separate systems depending on layout, risk, and validation requirements.
What should buyers check before choosing a checkweigher?
Buyers should review pack size, target weight, line speed, dynamic accuracy, conveyor stability, reject method, data records, cleaning access, and integration with upstream and downstream machines.
Referencje
- FDA / eCFR — 21 CFR Part 11, Electronic Records; Electronic Signatures.
https://www.ecfr.gov/current/title-21/chapter-I/subchapter-A/part-11
- European Commission — EU GMP Annex 11: Computerised Systems.
https://health.ec.europa.eu/system/files/2016-11/annex11_01-2011_en_0.pdf


