Intro
What exactly is cGMP?
Every time you unpack products in our daily life, you may wonder how these products stay safe, estável, and trustworthy batch after batch?
The answer is: cGMP. Short for Current Good Manufacturing Practice, a rigorous standard governing the full production from manufacturing and shipment, ensuring the safety, consistency, and quality of your healthcare and skincare products.
Learn more about cGMP, its safety, trustworthiness and certification here. Enjoy your read!
1. Definição
1.1 What is cGMP?

Fig 1: Ruidapacking SGS Certification – in Accordance with cGMP
cGMP is not merely an “enhanced GMP”, but rather, a “dynamic living quality standard”.
People mix them up all the time. But the core difference between them is like the distinction between a cook following a fixed recipe and a chef who truly masters the science behind cooking.
Boas Práticas de Fabricação (BPF) stands for Good Manufacturing Practice, laying out basic production requirements including facility hygiene and complete documentation. The “c” in “cGMP” means “current”, meaning that cGMP is not a set of fixed rules, but a living and adaptive framework which evolves alongside technology development and industry expertise.
1.2 cGMP vs. GMP

Fig 2: cGMP vs. GMP
The core difference between GMP and cGMP lies in their nature, requirements and implementation concepts.
GMP (Good Manufacturing Practice) is a set of basic and fixed production quality standards that mainly specifies basic requirements for production links, including production environment hygiene, personnel operation standards and record integrity. It aims to meet the minimum quality standards and guarantee basic product safety.
The “c” in “cGMP” denotes “current”. Its essence is a dynamic and adaptive management system that evolves alongside technological progress and industry demands. Unlike GMP’s rigid regulations, cGMP does not restrict to specific equipment models or operating steps, but emphasizes the practical effect of quality control.
As an example, GMP may specify the mixer model, while cGMP focuses more on whether the mixer can achieve uniform material mixing and meet quality requirements.
Besides, cGMP focuses on proactive risk prevention and full-process traceability, including strict raw material inspection, complete and traceable production data, and independent quality review. It is more comprehensive and strict than GMP, and is widely recognized as the core quality standard for the global pharmaceutical, food and cosmetic industries.
2. Ruidapacking and cGMP 4Q
As a professional equipment manufacturer, Ruidapacking’s products are deeply tied to cGMP compliance. Professional equipment like máquinas de enchimento de cápsulas, máquinas de embalagem em blister, prensas para comprimidos, máquinas de contagem, e máquinas de cartonagem (for pharmaceutical and nutraceutical purposes only) – is engineered for full cGMP compliant. The key to comply with cGMP requirements lies in 4Q – DQ(Design Qualification), QI(Qualificação de Instalação), QO(Qualificação Operacional), QP(Qualificação de Desempenho).

Fig 3: Ruidapacking Quality Management System Certificate
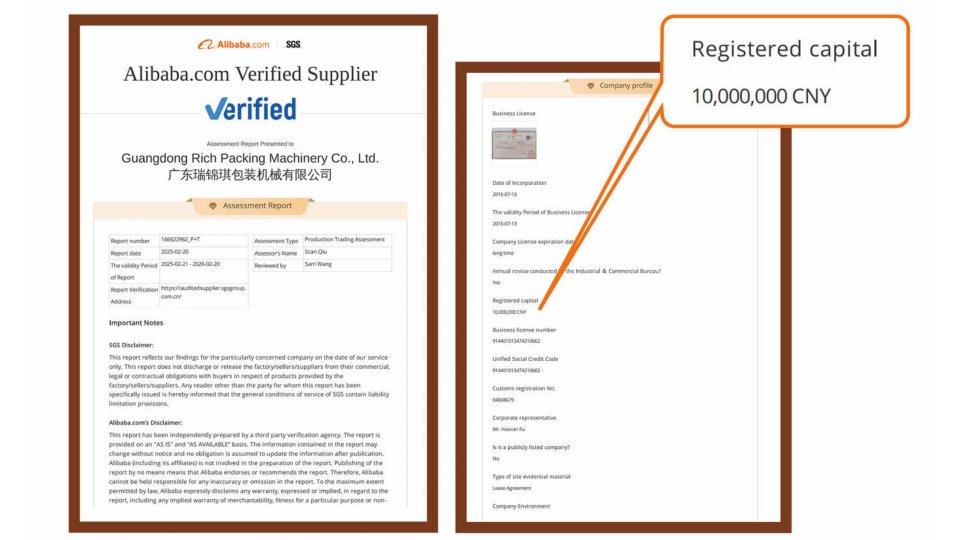
Fig 4: RuidApacking – Alibaba Verified Supplier
2.1 Comprehensive Validation Framework 4Q Qualification
- DQ: Design Qualification
Ruidapacking provides documented evidence that every machine design fully complies with pharmaceutical clients’ User Requirement Specification (URS) and all applicable cGMP standards.
- QI: Qualificação de Instalação
Proof that each Ruidapacking’s machine is installed accurately and completely in accordance with our specifications and design criteria.
- QO: Qualificação Operacional
Validation that our equipment functions reliably within its rated operating parameters during no-load or simulated load testing.
- QP: Qualificação de Desempenho
The most vital stage, verifying that Ruidapacking’s equipment delivers consistent, high-quality products when running with real production materials.
2.2 RuidApacking: 15+ Years of Expertise in cGMP-Compliant Professional Equipment Design and Performance
With over 15 years of industry experience, Ruidapacking designs professional equipment fully aligned with cGMP requirements. In structural design, our machines feature smooth rounded transitions without dead corners. Além disso, our machines use 316L stainless steel for contact surfaces for hygiene, minimizing residue. It also allows for quick disassembly, cleaning and disinfection, reducing human intervention, lowering production risks, in full conformity with cGMP’s end-to-end control.

Fig 5: RuidApacking – cGMP Compliant Manufacturer
Sealing and dust containment are configured to comply with clean-room specifications, providing an effective sterile and hygienic production environment. Regarding process control and data management, Ruidapacking uses electronic recording, user access control, and audit trails to satisfy cGMP criteria for data integrity.

Fig 6: RuidApacking – cGMP-Compliant Professional Equipment Design and Performance
In terms of operational stability, precise control of pressure, filling volume, and sealing ensures consistent quality of each batch, and truly achieves cGMP’s objective of predictable and reliable compliant. Hence, cGMP compliant equipment!
3. cGMP Full-Chain Protection from Source to Shelf

Fig 7: Ruidapacking Meets cGMP Standards – Global Certification Received
Quality by Design: at the heart of cGMP, there lies QbD – Quality by Design. It goes far beyond production, but extends chain protection to the entire life-cycle from research and development to market withdrawal. This system is like a sophisticated framework with strict standards at every stage.
3.1 cGMP – Raw Material Incoming
Raw Material Incoming: Inspection for every batch
Whether it is pharmaceutical or skincare active ingredient, cGMP demands a comprehensive supplier evaluation system. Upon arrival, all incoming raw material is inspected multiple times to verify conformity with registration standards.
A European pharmaceutical company was trying to enter the U.S. market, but was rejected by FDA because of inadequate raw material inspection record. Even the finished products passed inspection, it was disqualified because of insufficient process transparency.
3.2 cGMP Production Process: Data Integrity

Fig 8: Use ALCOA+ Criteria for Data Integrity
Processo de fabricação: Data speaks louder
Data integrity is one of cGMP’s top priorities.
According to EUROTHERM, The famous ALCOA+ principles define best practice guidelines: Attributable, Legible, Contemporaneous, Original, and Accurate; as well as complete, consistent, and traceable.
Modern manufacturers use computerized system to track production data live: temperatura, humidity, mixing time, etc.. If any value deviates from the standard, it triggers the alarm automatically, making it more effective than manual sampling and inspection.
RuidApacking's cGMP compliant equipment ensures full process traceability and validation throughout production.
3.3 cGMP Product Release
Product Release: the qualified person has full veto power
Pursuant to China’s cGMP requirements, enterprises must establish an independent quality management unit. The qualified person carries out the final review for each batch and holds full veto power. Products can only be signed off for release when the entire production and inspection process complies with the standards. Any incomplete documentation, the products are prohibited from being marketed.
4. Why is cGMP Regarded as the International Passport?
4.1 “Current” as the Core Principle
Within the global marketplace, cGMP has long been the universal language representing product quality. Though different countries vary in their regulatory systems, the core principles remain largely aligned.
cGMP in the U.S. FDA is famous for its dynamic inspections. Unannounced random checks are conducted base on each enterprise’s compliance records and product risk level. It has zero tolerance for data manipulation. An international pharmaceutical company (Ranbaxy) faced significant compliance issues and was fined billions of dollars for deleting disqualified data.(from Wikipedia)
EU GMP underscores full employee participation. It requires everyone, from senior managers to operational personnel, to have clear quality responsibility. It further features the system of Quality Persons who are legally accountable for product quality.
4.2 cGMP Global Passport

Fig 9: Ruidapacking Cartoning Machine Workshop
China’s 2010 GMP released by NMPA has extensively incorporated cGMP concepts. Through regular unannounced inspections, it has strengthened supervision. Enquanto isso, it has issued specific rules for local industries such as Chinese medicine, achieving a balance between domestic adaptation and global alignment.
For enterprises, complying cGMP requirements means obtaining the “cGMP Global Passport” in the international market. Ruidapacking exports pharmaceutical packaging equipment to over 85 países. This is made possible by its compliance with ISO, CE, FDA and approval from other international authorities. Behind all these registrations and certifications lies the company’s full adherence to cGMP requirements.
5. cGMP in Our Daily Life and Industry Applications
Many people think that cGMP is exclusive to pharmaceutical industries. But the truth is, its scope has long extended to industries like:
- Professional equipment like what Ruidapacking manufactures (máquinas de enchimento de cápsulas, prensas para comprimidos, alu-plastic blister packaging machines, máquinas de contagem, cartoning machines for pharmaceutical, nutracêutico, chemical and daily products);
- Nutraceutical and supplement production lines;
- Cosmetics, skincare, and personal care;
- Comida, function food, infant formula;
- Vaccines, bio-pharmaceuticals, and medical consumables.
These unseen safeguards ultimately translate into predictable quality and consistency. The active ingredient in each product (for pharmaceutical, nutraceutical and skincare purposes only) is safe and stable, and every use is free from risks caused by batch to batch variations.
Conclusão
cGMP is like a commitment to trust and reliability.
Since the introduction of ICH Q7A guideline in 1997 to today’s digitized and intelligent quality control, the cGMP has always centered on one goal – turning product quality from accidental compliance to inevitable compliance.
Behind this tiny acronym are comprehensive risk control. Além disso, there are also adaptive technologies and an unwavering promise to safe and effective use. This is the greatest value of cGMP.