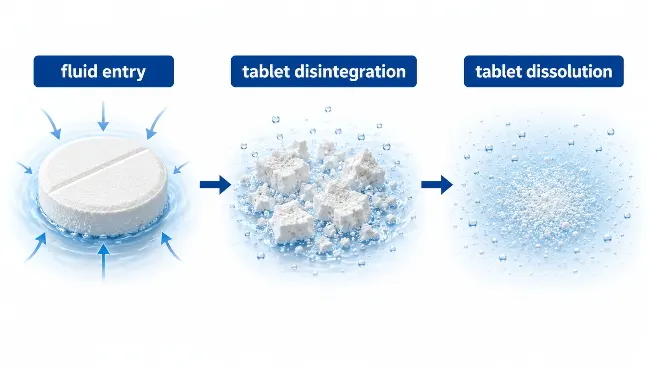
Come si dissolve una compressa? Nella maggior parte dei casi, una compressa non si scioglie semplicemente. Innanzitutto assorbe liquidi, inizia a rompersi attraverso la disintegrazione della compressa, e quindi espone più area superficiale per la dissoluzione della compressa. Questo è il motivo per cui due compresse dissolventi possono comportarsi diversamente nello stesso fluido: uno potrebbe rompersi rapidamente, mentre un altro può mantenere la sua forma più a lungo.
Durezza della compressa, rivestimento della compressa, progettazione della formulazione, esposizione all'umidità, e l'imballaggio dei tablet influenzano tutti questo processo. Una compressa a rapida dissoluzione, una compressa non rivestita, una compressa ricoperta di zucchero, e una compressa a rivestimento enterico non sono progettate per degradarsi allo stesso modo.
Per i produttori di tablet, marche di integratori, e team di confezionamento, questa domanda diventa pratica. La forza di compressione della compressa influisce sulla durezza della compressa. Il rivestimento della compressa può proteggere la superficie, gusto della maschera, o ritardare il rilascio. La confezione delle compresse protegge il prodotto finito prima dell'uso, soprattutto quando l'umidità, danni al rivestimento della compressa, sigillatura delle bottiglie, o le prestazioni della barriera anti-blister possono influenzare la stabilità della compressa. Comprendere come una compressa si dissolve aiuta a collegare la formulazione della compressa, compressione della compressa, rivestimento della compressa, test delle compresse, e il confezionamento dei tablet in un'unica decisione di produzione.

Come si scioglie una compressa dopo aver ingerito del liquido?
Una compressa compressa standard solitamente si dissolve attraverso una sequenza. Il fluido raggiunge prima la superficie della compressa, poi si sposta nei pori, crepe, o parti più deboli del patto. Man mano che la struttura si indebolisce, la disintegrazione della compressa crea frammenti più piccoli. Una superficie più esposta consente quindi di continuare la dissoluzione della compressa.
La dissoluzione delle compresse non segue tutte le stesse tempistiche. Le compresse non rivestite possono consentire al fluido di raggiungere il nucleo della compressa più velocemente. Le compresse rivestite aggiungono uno strato esterno che deve interagire con il fluido prima che la struttura interna si apra. Una compressa a dissoluzione rapida o una compressa orale dispersibile è progettata per una disintegrazione più rapida della compressa, mentre una compressa a rilascio modificato o una compressa con rivestimento enterico segue una strategia di rilascio diversa.
| Termine | Significato semplice | Collegamento di produzione |
| Disintegrazione della compressa | La compressa si rompe in pezzi più piccoli | Influenzato dalla forza di compressione della compressa, eccipienti, rivestimento della compressa, e umidità |
| Dissoluzione della compressa | Gli ingredienti si dissolvono nel liquido | Utilizzato per valutare il comportamento di rilascio e la coerenza dei batch |
| Durezza della compressa | Resistenza alla rottura o allo schiacciamento delle compresse | Collegato alla forza di compressione della compressa e alla forza di manipolazione |
| Rivestimento della compressa | Strato esterno sulla tavoletta | Può proteggere, gusto della maschera, migliorare l'aspetto, o rilascio del controllo |
| Test di disintegrazione per compresse | Controlla la rottura della compressa in condizioni definite | Controllo di qualità comune per compresse e capsule |
| Test di dissoluzione per compresse | Misura il rilascio della compressa nel mezzo di prova | Importante nello sviluppo del prodotto e nel controllo qualità |
Disintegrazione della compressa vs dissoluzione della compressa
La disintegrazione e la dissoluzione della compressa sono strettamente correlate, ma misurano aspetti diversi delle prestazioni del tablet. La disintegrazione delle compresse chiede se la forma farmaceutica si scompone in pezzi più piccoli. La dissoluzione della compressa chiede quanto ingrediente entra in soluzione nel tempo.
Un test di disintegrazione delle compresse utilizza attrezzature e condizioni definite per verificare se le compresse si rompono entro il tempo richiesto. Questo tipo di test sulle compresse è comune nel controllo di qualità di compresse e capsule, soprattutto quando la rottura prevedibile fa parte della progettazione del prodotto.
Un test di dissoluzione per compresse utilizza una configurazione diversa. Misura il rilascio degli ingredienti in un mezzo di prova nel tempo, che aiuta i team di formulazione e quelli di qualità a confrontare le prestazioni delle compresse in condizioni controllate.
Per gli acquirenti di attrezzature, il valore è pratico. Compressione della compressa, rivestimento della compressa, manipolazione delle compresse, e la confezione delle compresse non deve creare variazioni che verranno successivamente visualizzate nei risultati del controllo qualità. Una linea di produzione stabile non può sostituire il lavoro di formulazione, ma può ridurre i danni evitabili e la variazione della compressa dopo che il prodotto ha lasciato la comprimitrice.
In che modo la durezza delle compresse influisce sulla dissoluzione delle compresse
La durezza delle compresse è uno dei collegamenti più chiari tra la produzione e il comportamento delle compresse. Una compressa troppo morbida potrebbe scheggiarsi, berretto, laminato, o perdere polvere durante il trasferimento. Una compressa troppo dura potrebbe resistere alla penetrazione dei liquidi e rompersi più lentamente del previsto. Questo è uno dei motivi per cui una compressa si dissolve in modo diverso quando viene applicata la forza di compressione della compressa, durezza della compressa, o cambiamenti nella porosità della compressa.
La durezza della compressa è influenzata principalmente dalla formulazione, qualità dei granuli, profondità di riempimento, precompressione della compressa, compressione della compressa principale, e tempo di sosta. UN comprimitrice rotativa o la macchina comprimitrice deve produrre compresse di peso stabile, spessore della compressa, e la durezza della compressa in tutto il lotto. La sola velocità della comprimitrice non è sufficiente se le compresse finite diventano fragili o troppo dense.
I laboratori utilizzano spesso un tester per la durezza delle compresse, tester per compresse, o durometro per compresse per verificare la resistenza meccanica. Durometri digitali a tavoletta, durometri automatici per compresse, e i durometri per compresse portatili vengono utilizzati in diverse impostazioni di controllo qualità, a seconda della routine di test dei tablet e della scala di produzione.
La durezza delle compresse influisce anche sul confezionamento delle compresse a valle. Le compresse molli possono rompersi in una linea di conteggio e imbottigliamento delle compresse. Le compresse fragili possono scheggiarsi durante l'inserimento del blister. Le compresse eccessivamente dure possono sopravvivere alla manipolazione ma creare preoccupazioni nel comportamento di disintegrazione delle compresse.
Come il rivestimento della compressa modifica la dissoluzione della compressa
Il rivestimento della compressa modifica il modo in cui la superficie della compressa incontra il fluido, aria, e umidità. Può anche cambiare il modo in cui una compressa si dissolve perché il liquido deve prima interagire con lo strato esterno prima di raggiungere il nucleo della compressa. Alcuni rivestimenti delle compresse sono principalmente protettivi o visivi. Altri sono funzionali e progettati per cambiare il luogo o il modo in cui inizia il rilascio dei tablet.
I tipi comuni di rivestimento delle compresse includono il rivestimento con pellicola, rivestimento di zucchero, rivestimento enterico, e modelli di compresse rivestite compresse. Le compresse rivestite con film utilizzano uno strato relativamente sottile che può migliorare l'aspetto, ridurre la polvere, gusto della maschera, e facilitano la deglutizione. Il loro sottile strato esterno può modificare la velocità con cui il fluido bagna la superficie della compressa, ma l'effetto principale dipende dalla formula del rivestimento, spessore del rivestimento della compressa, e uniformità del rivestimento della compressa.
Una compressa rivestita di zucchero ha uno strato esterno più spesso. Le pillole ricoperte di zucchero possono migliorare il gusto e l'aspetto, ma lo spessore aggiunto può modificare le dimensioni della compressa. Ciò può influire sul design della cavità della vescica, conteggio del riempimento della bottiglia, e flusso di conteggio delle compresse. Anche il rivestimento con zucchero e il rivestimento con pellicola impongono requisiti diversi in termini di tempo di rivestimento delle compresse, qualità della superficie della compressa, e la gestione a valle dei tablet.
Le compresse con rivestimento enterico utilizzano un rivestimento progettato per resistere a condizioni acide e rilasciato successivamente in diverse condizioni di pH. Questo tipo di rivestimento delle compresse richiede un attento controllo del processo poiché lo strato di rivestimento funzionale delle compresse deve rimanere intatto attraverso il confezionamento delle compresse, magazzinaggio, e trasporti.
Anche il materiale di rivestimento della compressa influisce sulle prestazioni. Rivestimento per compresse HPMC, rivestimento con film di ipromellosa, sistemi di rivestimento acquosi, materiali di rivestimento di pellicole per compresse, e i materiali di rivestimento enterico indicano tutti una preoccupazione dell’acquirente: lo strato di rivestimento della compressa deve rimanere consistente e stabile.
Dissoluzione rapida, ODT, e compresse non rivestite
Una compressa a dissoluzione rapida è progettata per rompersi rapidamente rispetto a una normale compressa compressa. Una compressa orale dispersibile o compressa ODT appartiene a un gruppo di prodotti correlato. Questi formati spesso richiedono un imballaggio accurato delle compresse perché la struttura delle compresse è più sensibile all'umidità e può rompersi rapidamente.
Le compresse non rivestite sono più semplici dall'esterno, ma non sono automaticamente più facili da produrre o confezionare. Una compressa non rivestita potrebbe esporre il nucleo della compressa più velocemente, ma potrebbe anche creare più polvere o evidenziare più chiaramente i difetti superficiali.
Le compresse rivestite e non rivestite differiscono anche nella manipolazione delle compresse. Se le guide possono causare danni al rivestimento della compressa, pennelli, tracce, o le parti di alimentazione sono ruvide. Le compresse non rivestite potrebbero richiedere una depolverazione più forte prima della sigillatura del blister o del confezionamento del flacone.
In che modo i fattori di imballaggio delle compresse influiscono sulla stabilità prima dell'uso
La confezione delle compresse non decide direttamente come una compressa si dissolve dopo l'uso. Protegge il tablet prima dell'uso, contribuendo a preservare la durezza della compressa, integrità del rivestimento della compressa, e livello di umidità. Tale protezione influisce sul fatto che la forma farmaceutica raggiunga l'utilizzatore nelle condizioni previste dal lavoro di formulazione e di test delle compresse.
L’esposizione all’umidità è uno dei rischi principali. Alcune compresse si ammorbidiscono, rigonfiamento, perdere la qualità della superficie, o diventare più fragili dopo aver assorbito l'umidità. Compresse a rapida dissoluzione, compresse effervescenti, e alcune compresse rivestite possono essere particolarmente sensibili. Materiale barriera anti-vesciche, sigillatura delle bottiglie, inserimento dell'essiccante, e la qualità della chiusura aiutano a ridurre tale esposizione.
La confezione in blister protegge le compresse in tasche separate. Questo formato è adatto alle compresse che necessitano di protezione per dose unitaria o di una migliore separazione dell'umidità tra le dosi. Una macchina confezionatrice blister deve corrispondere alle dimensioni della compressa, forma di compressa, forza del rivestimento della compressa, e comportamento alimentare.
L'imballaggio delle bottiglie supporta linee di compresse e integratori ad alto volume. Una linea di conteggio e imbottigliamento delle compresse può essere efficiente per conteggi più grandi, ma la bottiglia viene aperta più volte dopo l'acquisto. Inserimento dell'essiccante, sigillatura ad induzione, controllo della macchina tappatrice per bottiglie, e l'accuratezza dell'etichettatura diventano parte del piano di stabilità del tablet.
Quali apparecchiature supportano la dissoluzione delle compresse e la qualità del confezionamento?
Il percorso dell'attrezzatura non sostituisce il lavoro di formulazione, ma può proteggere le condizioni previste del tablet. Una comprimitrice rotativa forma il compatto, UN macchina per rivestimento di compresse applica e asciuga lo strato di rivestimento della compressa, e un depolveratore per compresse rimuove la polvere sciolta prima del confezionamento. Dopo di che, la linea solitamente segue uno dei due percorsi. In un percorso di confezionamento in blister, le compresse vengono inserite nelle cavità del blister, sigillato, e poi trasferito ad una astucciatrice. In un percorso di confezionamento delle bottiglie, le compresse passano attraverso il conteggio delle compresse, riempimento delle bottiglie, tappatura della bottiglia, sigillatura ad induzione, etichettatura, e poi l'astucciamento se è richiesto un imballaggio secondario.
Per tablet legati a prestazioni sensibili alla dissoluzione, tre controlli dell'attrezzatura meritano attenzione. Il controllo della compressione della compressa mantiene il peso della compressa, spessore della compressa, e la durezza della compressa è stabile. Il controllo del rivestimento della compressa supporta uno spruzzo uniforme, essiccazione, movimento della padella, e ripetibilità. Il trasferimento delicato della confezione delle compresse riduce l'altezza di caduta, attrito, e vibrazioni durante l'alimentazione del blister, conteggio delle bottiglie, e astucciamento.
Ruidapacking supporta questa visione dell'intero processo testando le apparecchiature prima della consegna con funzionamento continuo 24 ore su 24 e verifica a carico elevato di 8 ore per una movimentazione stabile dalla produzione delle compresse al confezionamento finale.
Come i produttori possono collegare la dissoluzione dei tablet, Rivestimento, e imballaggio
I produttori non scelgono la confezione dei tablet solo in base alle dimensioni del tablet o alle preferenze del mercato. La forma di dosaggio dovrebbe guidare il percorso.
Un duro, stabile, la compressa non rivestita può funzionare bene durante il conteggio e l'imbottigliamento ad alta velocità. Una compressa sensibile all'umidità a dissoluzione rapida potrebbe richiedere una protezione barriera più forte. Una compressa rivestita di zucchero potrebbe richiedere più spazio nella cavità del blister a causa del suo strato esterno più spesso. Una compressa con rivestimento enterico può richiedere un'attenta manipolazione per proteggere il rivestimento funzionale della compressa.
Un flusso di lavoro pratico è semplice: definire il tipo di rilascio della compressa e la sensibilità della compressa, confermare la durezza della compressa, friabilità della compressa, forza del rivestimento della compressa, e il comportamento dell'umidità, quindi scegliere un percorso di confezionamento delle compresse che protegga la forma di dosaggio.
| Condizioni della tavoletta | Preoccupazione produttiva | Preoccupazione per l'imballaggio |
| Compressa morbida o friabile | Rottura della compressa durante il trasferimento | Conteggio delicato delle compresse, depolverazione della compressa, e altezza di caduta ridotta |
| Compressa rivestita con film | Sfregamento della compressa o usura del rivestimento della compressa | Alimentazione regolare e adattamento corretto alla cavità della vescica |
| Compressa ricoperta di zucchero | Tablet di dimensioni maggiori e danni alla superficie | Correggere la dimensione della cavità o la pianificazione del conteggio delle bottiglie |
| Compressa con rivestimento enterico | Integrità funzionale del rivestimento della compressa | Confezione barriera e manipolazione attenta delle compresse |
| Compressa a rapida dissoluzione | Sensibilità all'umidità | Forte protezione dall'umidità e sigillatura |
| Compressa non rivestita | Polvere e superficie esposta della compressa | Depolverazione delle compresse, sigillatura, o protezione per dose unitaria |
Conclusione
Capire come si scioglie una compressa significa guardare oltre la semplice risposta “veloce o lento”.. Le compresse di solito si disintegrano per prime, quindi gli ingredienti si dissolvono. La durezza della compressa influisce sul modo in cui il fluido entra nel compatto. Il rivestimento delle compresse modifica il modo in cui la superficie interagisce con l'umidità e i mezzi di prova. I fattori di confezionamento delle compresse proteggono la forma di dosaggio finita prima che raggiunga l'utente.
Per gli acquirenti di attrezzature, questo crea un modo utile per confrontare i percorsi di produzione. La macchina comprimitrice, macchina per il rivestimento di compresse, macchina confezionatrice blister, linea di conteggio e imbottigliamento delle compresse, tappatrice per bottiglie, macchina sigillatrice ad induzione, etichettatura, e l'astucciatrice dovrebbe proteggere le condizioni progettate della compressa.
Domande frequenti
-
Come si dissolve una compressa?
Una compressa di solito assorbe prima i liquidi, poi si rompe in pezzi più piccoli attraverso la disintegrazione delle compresse. Dopo di che, viene esposta una maggiore superficie, e la dissoluzione della compressa può continuare.
-
Qual è la differenza tra disintegrazione e dissoluzione della compressa??
La disintegrazione della compressa significa che la compressa si rompe in pezzi più piccoli. La dissoluzione della compressa significa che gli ingredienti solubili si spostano nel liquido. Entrambi sono imparentati, ma misurano aspetti diversi delle prestazioni del tablet.
-
La durezza della compressa influisce sul modo in cui una compressa si dissolve?
SÌ. La durezza della compressa può influenzare la velocità con cui il fluido entra nella compressa. Una compressa troppo morbida potrebbe rompersi durante la manipolazione, mentre una compressa troppo dura potrebbe rompersi più lentamente del previsto.
-
Il rivestimento della compressa modifica la dissoluzione della compressa?
SÌ, a seconda del tipo di rivestimento della compressa. Il rivestimento della pellicola può influenzare principalmente la bagnatura e la protezione della superficie. Il rivestimento enterico è progettato per il rilascio ritardato in condizioni specifiche. Il rivestimento di zucchero modifica le dimensioni della compressa, gusto, e comportamento della superficie.
-
Cos'è un test di disintegrazione per compresse?
Un test di disintegrazione delle compresse verifica se le compresse si rompono in condizioni definite. È comunemente usato nel controllo qualità di compresse e capsule.
-
Cos'è un test di dissoluzione per compresse?
Un test di dissoluzione per compresse misura il rilascio degli ingredienti in un mezzo di prova nel tempo. Viene utilizzato nel lavoro di formulazione di compresse, controllo di qualità, e confronto dei prodotti.
-
Le compresse a dissoluzione rapida sono identiche alle compresse ODT?
Sono termini di prodotto strettamente correlati, ma le definizioni esatte possono dipendere dagli standard di prodotto e dalla formulazione del mercato. Generalmente, entrambi descrivono compresse progettate per scomporsi più velocemente delle normali compresse da deglutire.
-
La confezione delle compresse può influenzare il modo in cui una compressa si dissolve??
La confezione delle compresse non controlla direttamente la dissoluzione dopo la deglutizione. Protegge le compresse prima dell'uso, quindi umidità, danni al rivestimento della compressa, o un trattamento brusco non modificano le condizioni del tablet prima che raggiunga l’utente.
-
Per le compresse è meglio l'imballaggio in blister o in bottiglia?
Dipende dalla sensibilità del tablet, dimensione del conteggio, obiettivo di durata di conservazione, costo, e formato di mercato. La confezione in blister offre protezione per dose unitaria. L'imballaggio delle bottiglie può essere efficiente per conteggi più grandi, ma potrebbe essere necessario l'inserimento di un essiccante, sigillatura ad induzione, e un forte controllo della tappatura delle bottiglie.
Riferimenti
USP – Capitolo Generale <711> Dissoluzione
https://www.usp.org/sites/default/files/usp/document/harmonization/gen-method/stage_6_monograph_25_feb_2011.pdf





















