Einführung
Unit dose packaging places each prepared dose in its own sealed presentation. In Pharma- und Medikamentenverpackungen, das bedeutet normalerweise eine Tablette, eine Kapsel, eine Pulverpackung, oder ein Einweg-Flüssigkeitsbehälter, der so angeordnet ist, dass er identifiziert werden kann, geschützt, und als eine Verwaltungseinheit genutzt. FDA treats drugs packaged in unit dose containers as a distinct labeling case for oral solid and liquid dosage forms, which shows this is more than just a small pack format.
This packaging approach matters because its role goes beyond simple product containment. It can support dose-level identification, reduce repeated handling, and help maintain clearer control over each individual dose from packaging through dispensing and use. That is why unit dose packaging often appears in discussions about blister packs, strip packs, Beutel, and single-use liquid containers, especially in pharmaceutical manufacturing, hospital pharmacy, and medication-use settings.

What Is Unit Dose Packaging in Pharmaceuticals?
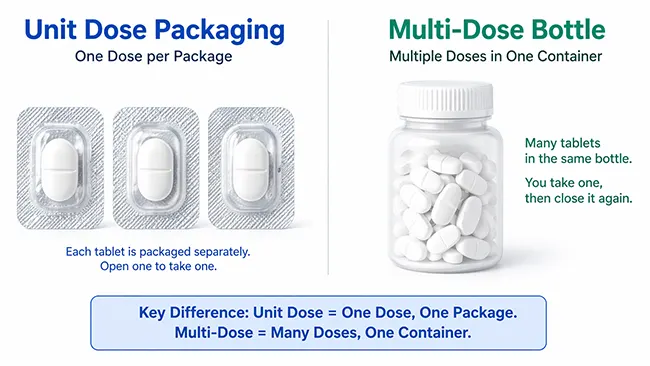
Unit dose packaging means a drug product is packed so one discrete dose is presented in its own package and handled as a single administration unit.
A simple way to understand it is this: the package boundary follows the dose. A bottle with 30 tablets may be compact, but it is still a multi-dose container because the user opens one pack and removes doses one by one. A blister pack with one tablet in each cavity, a strip pack with one dose sealed between flexible webs, or a single-use liquid container already separates each dose before dispensing or administration.
That difference affects protection, identification, and workflow. When each dose is individually separated, the package can make dose-level information clearer, limit repeated exposure of the remaining product after opening, and support more controlled downstream handling. This is one reason unit dose packaging is widely used where product protection, organization, and traceability matter alongside convenience.
In der Praxis, unit dose packaging can appear in several forms. Oral solid products are often presented in blister or strip formats. Powders may use small packets or sachets. Certain liquid products use single-dose containers. The format can change, but the core idea stays the same: one prepared dose, one defined package, one clearer point of use.
Unit Dose Packaging vs Multi-Dose Packaging
The most direct way to understand the difference is to examine the level at which the packaging separates and presents the dose. In a unit-dose format, each dose is physically separated before dispensing or administration. In a multi-dose format, several doses remain inside one container and are taken out over time. That difference shapes labeling, product exposure after opening, and day-to-day handling.
Unit-dose formats usually provide stronger dose-level separation. Each cavity, Beutel, or container stays sealed until use. Multi-dose formats trade that separation for simpler bulk storage and lower packaging intensity. Neither route is automatically better. They solve different problems.
For many oral solids, the decision is practical. If the product benefits from clearer dose presentation, more controlled handling, or more visible unit-level identification, a unit-dose route makes sense. If the product is routinely dispensed from one container and dose-by-dose separation adds little value, a multi-dose format may still be the better fit.
| Aspekt | Unit dose packaging | Multi-dose packaging |
| Dose presentation | One prepared dose per package | Multiple doses in one container |
| Handling after opening | Unused doses remain sealed | Remaining doses are exposed after each opening |
| Dose-level labeling | Stronger | Usually relies more on the outer label |
| Traceability | Easier at the individual-dose level | Usually managed at the container level |
| Packaging material use | Höher | Untere |
| Beste Passform | High-control, dose-specific use | Repeated dispensing from one container |
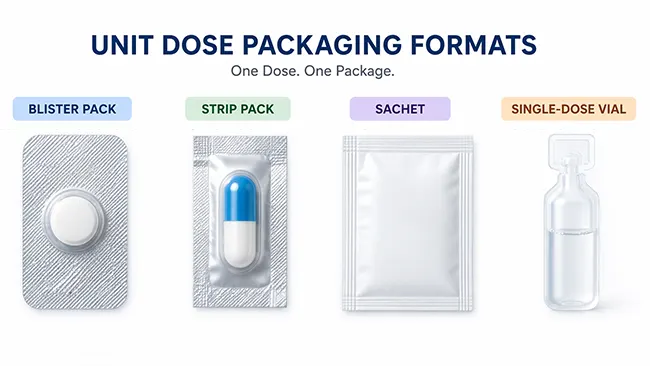
Common Types of Unit Dose Packaging
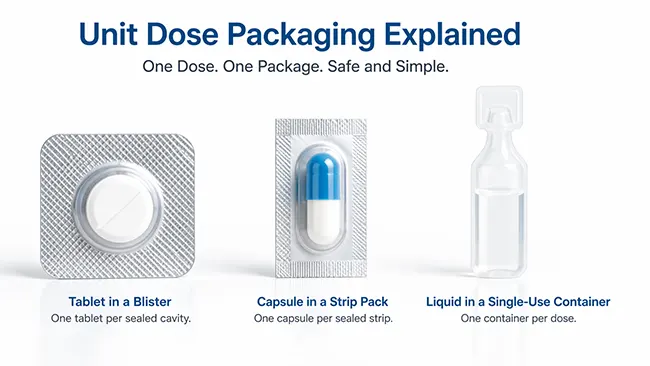
For tablets and capsules, Blisterpackungen are usually the first format people think of. Each cavity holds one dosage unit, so the product stays separated until the moment of use. This format supports visual organization and helps protect unopened doses from the repeated exposure that happens once a bottle is opened.
Strip packs follow the same basic logic. Instead of a formed cavity with lidding foil, the dose is sealed between flexible webs. Structurally, the package looks different from a blister, but functionally it still works as a unit-dose presentation because each dose is already separated before dispensing or administration.
Powders and granules may use small packets or sachets when the product is intended for one-time use. Certain liquid medicines use single-dose cups, Fläschchen, or ampoules. The product form changes, but the packaging principle does not: one prepared dose is already set apart inside its own package.
The most useful way to think about format choice is not to ask which package looks most familiar. It is to ask which package best matches the dosage form, Barrierebedürfnisse, labeling requirements, and downstream use pattern.
Why Unit Dose Packaging Is Used in Pharma and Healthcare
Vorteil 1: clearer dose-level identification
When each dose has its own package, identification becomes easier at the point of use. The package can be associated more directly with the product name, strength, and other handling information. That matters when the goal is not only to store the medicine, but also to keep each individual dose easier to recognize and manage.
Vorteil 2: less repeated handling of the remaining product
A multi-dose bottle is opened again and again over its service life. A blister, strip, or packet keeps unopened doses separated until needed. That can help reduce repeated exposure of the remaining product and support more controlled downstream handling.
Vorteil 3: stronger support for traceability
Because the dose is already separated, labeling and scanning can work at a smaller control point. This can make package-level tracking, dispensing control, and recordkeeping more straightforward, especially when the packaging workflow depends on clearer identification of each unit.
Vorteil 4: a better fit for organized dosing and sensitive products
Not every product needs unit-dose packaging, but it is useful when product protection, dose sequencing, or controlled presentation matter. That is one reason it remains common in oral solid packaging discussions, especially where Pharmaverpackungen has to balance protection, Benutzerfreundlichkeit, and workflow clarity.
What Are the Limits of Unit Dose Packaging?
The biggest trade-off is packaging intensity. Separating every dose usually means more material, more sealing steps, more printed area, and more unit-level inspection or coding work. Compared with a bottle or other bulk format, this can raise packaging complexity even when the product-level benefits are clear.
Cost is the second major issue. A unit-dose route may require more packaging material, tighter line control, and more attention to changeovers or printed information. That does not mean it is uneconomical. It means the format needs to be justified by product needs and operational priorities.
There is also a product-fit question. Some medicines work well in bottle or other multi-dose formats because the dispensing pattern, shelf-life profile, or commercial route does not require dose-by-dose separation. The existence of both unit-dose and multi-dose systems in current practice is the clearest sign that neither format is universally right.
Beschriftung, Barcodes, and Traceability in Unit Dose Formats
FDA guidance treats the unit dose container as a meaningful labeling surface for oral solid and liquid dosage forms, while ASHP guidance emphasizes clear identification, strength declaration, expiration dating, control information, and ease of safe use. That combination makes it clear that unit dose packaging is not just about dividing product into smaller packs. It is also about what each pack must communicate and support at the point of use.
In praktischer Hinsicht, the more value you place on dose-level visibility, scan-ready identification, and controlled downstream handling, the more attractive this format becomes. That is why unit dose packaging sits at the intersection of packaging design, compliance, and operational workflow rather than belonging to only one department.
What Packaging Equipment Is Used for Unit Dose Formats?
On the equipment side, oral solids most often reach this format through Blisterverpackungsmaschinen oder strip packaging machines. In a blister route, a machine forms or indexes the package, places the dose, seals the lidding layer, and creates a defined one-dose presentation in each cavity. In a strip route, the machine seals each dose between flexible materials while maintaining the same one-unit packaging logic.
Powders and granules may move through sachet packing machines when the product is intended for single use. Certain liquid products use single-dose cups, Fläschchen, or related filling routes depending on product form and administration logic.
The important point is that the equipment decision comes after the packaging decision. Erste, the manufacturer decides whether the product needs dose-by-dose separation. Then the line is chosen to deliver that format with the right barrier, Siegel, Codierung, and output stability.
Abschluss
Unit dose packaging is most accurately defined as a format centered on one prepared dose within one distinct package. Its value becomes clearer when dose-level identification, controlled handling, Rückverfolgbarkeit, and product protection matter more than the simplicity of a multi-dose container.
It is not automatically the right answer for every medicine. But it remains one of the most important format choices in pharmaceutical and medication packaging because it changes how the product is labeled, geschützt, handled, and used. In der Praxis, the best decision is rarely about choosing the “modern” option or the “traditional” one. It is about matching the packaging format to the product, the workflow, and the level of control the application really needs.
Häufig gestellte Fragen
Is unit dose packaging the same as single-dose packaging?
Nicht immer, but the two often overlap. In practical use, both ideas point to a package that presents one prepared dose at a time. The exact wording may differ by context, but the core packaging logic is usually similar.
Is blister packaging always unit dose packaging?
Blister packaging is commonly used for unit-dose presentation, especially for tablets and capsules, because each cavity can hold one dosage unit. But the key question is not the package name alone. It is whether the product is actually organized as separate prepared doses.
Is unit dose packaging better than multi-dose packaging?
Nicht in jedem Fall. Unit-dose packaging is stronger when dose-level control, identification, and handling matter most. Multi-dose packaging may still be the better commercial or operational choice when repeated dispensing from one container is practical and dose-by-dose separation adds little value.
What products commonly use unit-dose formats?
Common examples include tablets and capsules in blister or strip packs, powders in small packets or sachets, and certain liquids in single-dose cups, Fläschchen, or ampoules.
Why is labeling especially important in unit-dose packaging?
Because the package is carrying information at the dose level rather than relying only on a larger outer container. That makes label clarity more central to safe handling and downstream use.
Can unit dose packaging help with traceability?
Ja. Individually separated doses make it easier to connect the package to specific labeling, scanning, and recordkeeping steps.
Does unit dose packaging usually cost more?
It often can, because it usually requires more materials, more unit-level handling, and more packaging control than a multi-dose route. Whether that extra cost is justified depends on product needs and workflow priorities.
Referenzen
·ASHP — Single Unit and Unit Dose Packages of Drugs
https://www.ashp.org/-/media/assets/policy-guidelines/docs/technical-assistance-bulletins/technical-assistance-bulletins-single-unit-and-unit-dose-packages-drugs.pdf
·Manrex — Multi-Dose or Unit-Dose: How They Are Both Being Used in Long-Term Care
https://manrex.com/blog-post/multi-dose-or-unit-dose-how-they-are-both-being-used-in-long-term-care/


